
Wingspan
♂ 29 - 32 mm, ♀ 31 - 34 mm
Checklist Number
60.001
The Duke of Burgundy is the sole representative of a subfamily known as the "metalmarks", since some of its cousins, particularly those found in south America, have a metallic appearance. A curious characteristic of this subfamily is that the female has 6 fully-functional legs, whereas the male has only 4 - the forelegs being greatly reduced. The Duke of Burgundy was once classified as a fritillary, given the similarity with those fritillary species found in the British Isles. This butterfly is found mainly in central southern England, although scattered colonies are found elsewhere such as in the north of England in Cumbria and Yorkshire. This species is not found in Wales, Scotland, Ireland, the Isle of Man or the Channel Islands. Although relatively-large colonies exist, most colonies only contain around a dozen individuals at the peak of the flight season.
| Family: | Riodinidae | Grote, 1895 |
| Subfamily: | Riodininae | Grote, 1895 |
| Tribe: | Hamearini | Tutt, 1906 |
| Genus: | Hamearis | Hübner, [1819] |
| Subgenus: | ||
| Species: | lucina | (Linnaeus, 1758) |
Description to be completed.
The distribution data (2000-2009) has been made available through the generosity of Butterfly Conservation and any subspecies distribution is taken from Riley (2007). Click here to see the distribution of this species together with site information overlaid.
This butterfly was, in the past, primarily known as a woodland butterfly, where it fed on Primroses growing in dappled sunlight, with a number of colonies in chalk and limestone grassland. However, the cessation of coppicing in woodlands has had a marked effect on this species, with many woodland colonies dying out as a result. Primrose is used as the larval foodplant in woodland, whereas Cowslip is used on grassland.
Long-term distribution and population trends show that this butterfly is in serious decline. It is therefore a priority species for conservation efforts. It is thought that this species is unable to survive intensive grazing of chalk and limestone grasslands and that this is, at least, one possible cause of the decline. It is also unable to tolerate areas where the foodplant becomes too shaded by surrounding shrubs and grasses. A delicate balance therefore exists that requires specific site management to cater for this delightful little butterfly.
The table below shows the occurrence (distribution) and abundance (population) trends, using information from The State of the UK's Butterflies 2015 (Fox, 2015). Any UK BAP status is taken from the UK Biodiversity Action Plan (UK BAP) (2007 review).
1976-2014 (%) | 1976-2014 (%) | 2005-2014 (%) | 2005-2014 (%) | Click here to access the Biodiversity Action Plan (BAP). |  -84 -84 |  -42 -42 |  +3 +3 |  +67 +67 |
|---|
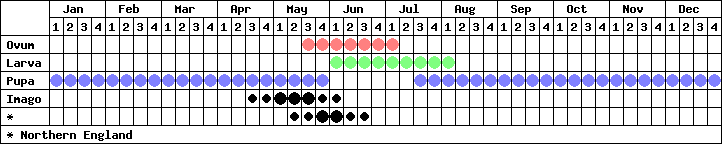
There is one brood each year, with the adults emerging at the end of April in southern sites, peaking in the middle of May. A partial second brood may appear in some years, but this is the exception, rather than the rule, and only occurs in certain sites in the south of England.
The sexes are similar in appearance, although the female tends to have more orange on the wings and rounder tips to the forewings. The male and female can also be distinguished by behaviour. The fast-flying males are extremely territorial and will sit on a favourite perch, darting out to inspect anything that might be a passing female. Once a virgin female is encountered, the two mate without any discernable courtship. This is usually in mid-morning just after the females have emerged. The flight of the female is not as rapid as the male and they are often seen when egg-laying as they move from plant to plant, landing on the edge of a leaf before curling their abdomen to lay on the underside of the leaf.
Adults only occasionally nectar, usually in warmer weather, with Wood Spurge, Buttercup, Hawthorn and Bugle being favourites. Both sexes roost in tall scrub or trees.
Adults feed primarily on buttercups (Ranunculus spp.), hawthorns (Crataegus spp.), Tormentil (Potentilla erecta) and Wood Spurge (Euphorbia amygdaloides).
Eggs are normally laid singly, or in small batches of just 3 or 4 eggs, on the underside of the edge of a leaf of the foodplant. Nearby foliage may be used on occasion, especially when the foodplant is within dense vegetation. Large, lush, green-leaved plants are typically used, either among grasses or close to scrub. Snails are known to cause heavy losses of eggs, as they feed on primula leaves during the spring. Eggs hatch in 1 to 3 weeks, depending on the weather.
"The eggs are deposited singly or in small clusters of four or five, but more often only in two or three, on the under surface of the leaves of primrose (Primula vulgaris) and cowslip (P. veris). Eggs laid by a captive female on May 27th, 1890, hatched on June 10th, remaining in the egg state fourteen days. A female captured on May 27th, 1901, deposited fifty-six eggs the following day on the under surface of the leaves of a potted-up primrose plant. These began hatching on June 9th, being twelve days in the egg state. Two captive females laid thirteen eggs on May 17th, 1911; the following three days were cold and sunless, consequently the butterflies remained motionless until the 21st, when thirty more eggs were laid, and during the following week about one hundred more were deposited. Those laid on the 17th hatched on May 31st, remaining fourteen days in the egg state. The egg is 0.60 mm. in diameter and about the same in height. It is almost globular in form, the micropyle is slightly sunken and the base somewhat flattened. Although the general appearance of the surface is highly polished, glistening and translucent, it is, when viewed under the microscope, found to be covered with an extremely delicate network of reddish-coloured reticulations, mostly hexagonal in pattern; appearing in the high light and encircling the egg close to the base are a number of extremely fine perpendicular ribs with knobbed apices. When first laid the egg is a very pale, translucent greenish-yellow-pearly-white colour, which remains unchanged until two days before hatching, when it becomes slightly more opaque, and finally the young larva is plainly visible through the glassy shell. The head produces an ochreous blotch at the crown, and the long dark hairs form a regular lattice-work diamond pattern round the egg; the colouring of the egg is then a light pearly-ochreous-greenish with opalescent reflections." - Frohawk (1924)
On hatching, the young larva eats its eggshell before moving to the base of the foodplant, feeding only at night. Larvae emerge at dusk, and can be found in torchlight, usually feeding on the upperside of the leaves. A tell-tale sign of a larva is a characteristic patchwork of holes made in the leaf surface, leaving the major veins intact. There are 4 instars in total and this stage lasts around 6 weeks.
The primary larval foodplants are Cowslip (Primula veris) and Primrose (Primula vulgaris). False Oxlip (Primula veris x vulgaris) is also used.
"The young larva makes its exit by eating away the whole of the crown of the egg, leaving a large circular hole. Directly after emergence it measures 1.6 mm. long. The head is rather large, of an ochre-yellow colour with a highly polished surface, and bears a number of fine glassy-white hairs, very dark eye spots, and reddish mouth parts. The colour of the body is pale primrose or creamy-yellow; it is profusely covered with long hairs. Those on the dorsal surface are remarkably formed; they consist of four on each segment, placed in pairs close together; the one nearest the centre of the back and on the anterior portion rises from a bulbous base in a simple stem and then bifurcates into long, sharply pointed, serrated prongs, both curving backwards, but the posterior one is much the longest. The second one, which is placed more sub-dorsally and more backwards, is of precisely similar construction, but both prongs are directed forwards; both pairs of prongs and stems are purplish with whitish bases. Close to the base of the first one is a very minute, sharp, spine-like hair which is only the height of the bulbous base. Close to and above the white spiracle is a long white slender hair. Immediately below the spiracle is a large bulbous lobe on which are four similar hairs, all diverging at their apices. On the ventral surface of each segment are three smaller hairs directed downwards; all the hairs are white, slender, slightly curved and serrated. The young larva feed on the under surface of the leaves, making numerous small perforations. Just before the first moult it measures 3.5 mm. long; usually the colouring is greyish-green, palest ventrally, but some are pale ochreous with only a slight tinge of green; all are translucent and rather shining." - Frohawk (1924)
"After the first moult, eleven days old, it measures 6.3 mm. long; the colour is a pale translucent grey-green, with a darker green medio-dorsal line; on either side of each segment is a cluster of black and white serrated hairs rising from olive-brown warts, forming a longitudinal sub-dorsal series. A few other hairs are scattered along the side, and a fringe of white hairs extends along the lateral line; all are serrated, but none are bifid as in the first stage. The head is ochreous with dark eye spots and mouth parts, and is beset with hairs." - Frohawk (1924)
"The second moult on June 15th, 1911. After the second moult, twenty-three days old, it measures 8.5 mm. long. The general colouring is pale olive-green, with ochreous sub-dorsal blotches, and pale grey-green (almost whitish) on the ventral surface. The head is deep ochreous or dull amber with dark eye spots, a medio-dorsal series of dull black spots, one on each segment, and a paler sub-dorsal series much less distinct. The body is now much more densely studded with black and white serrated hairs, and clothed also with extremely fine delicate wavy white hairs. The larva is particularly quiet in habits, resting motionless for hours at a time, mostly in a straight attitude on the under surface of the leaf of its food plant." - Frohawk (1924)
"The third moult on June 30th, 1911. After the third moult, fully grown, forty-three days old, it measures 15.9 mm. long. The body is rather stout in proportion, the back rather arched and tapering at each end, the segmental divisions indistinctly defined. It is clothed with numerous stiff, serrated bristles, both dark brown and pale ochreous, chiefly arranged in clusters on the dorsal and super-spiracular areas, and a sub-spiracular fringe projecting laterally, composed of four brown bristles and a dense cluster of whitish bristles, all serrated, on each segment; the entire surface is densely covered with extremely fine wavy white hairs. The head is light amber with a dark clypeus, eye spots and mouth parts, and beset with numerous bristles of various sizes similar to those on the body. The colour of the body is pale buff, slightly tinged with pale drab on the dorsal and lateral regions; a dark purplish-drab medio-dorsal line extends down the body, enclosing a black spot in the centre of each segment; along the sub-dorsal area is a series of oblique dull drab markings, one on each segment; the lateral stripe is pale ochreous, inclining to cream colour; the legs are ochreous and the claspers creamy-white. When the larva stops feeding and is ready for spinning up it turns a pale creamy-buff colour. The first one pupated July 13th, 1911, remaining forty-three days in the larval state." - Frohawk (1924)
The pupa is usually formed away from the foodplant in leaf litter, a grass tussock or other vegetation, secured by a silk girdle and the cremaster. It is believed that shrews are responsible for heavy losses during the pupal stage in which this species hibernates.
"The pupa measures 11 mm. long. The general form is short, stout and rounded. Dorsal view: Head rounded, with a slight central depression in front, slightly angular at base of wings, body swollen across the middle, abdomen conical, cremaster scarcely visible from above. Lateral view: Head rounded, thorax swollen, meta-thorax slightly sunken, abdomen swollen and rounded and strongly curved to anal segment, which terminates in a decurved cremaster furnished with long hooks; the ventral surface is flattened and straight. Excepting the last four abdominal segments the segmental and thoracic divisions are very indistinct. The ground colour is pale creamy-ochreous, with a delicate tinge of flesh colour over the head and wings. The thorax and abdomen are dotted with black, chiefly arranged in longitudinal rows, and two transverse bands of black on the head and along the inner margin of the wing; the neuration is indicated by fine lines. The surface, excepting the wings, is densely sprinkled with warts of various sizes, mostly very minute and amber-brown in colour; the largest are darker brown; all emit bristles of various lengths, those from the largest warts are of considerable length and thickly pectinated and all are amber-brown; the warts are in the form of flattened circular discs. The spiracles are ochreous and very indistinct. The pupa is attached by a cincture round the middle and the cremastral hooks to a pad of silk. All in captivity were spun upon the under side of the primrose leaves. At first the pupa is wholly cream coloured without markings, but the colouring soon matures." - Frohawk (1924)
The following links provide additional information on this butterfly.